Soil temperatures and fall ammonia application
As we indicated in a recent Bulletin article, nitrogen applied as anhydrous ammonia in the fall is an effective source of N for the corn crop, but is also a little more subject to loss compared to N applied in the spring. One of the main factors that determines how much of the fall-applied N is subject to loss is how much of the ammonia converts to nitrate (is nitrified) in the fall and early spring, before the next planting season.
One way to slow nitrification of fall-applied ammonia (which converts quickly to ammonium in the soil) is to add a nitrification inhibitor. The other way is to wait to apply ammonia until soil temperatures are low enough to limit the activity of the bacteria responsible for the conversion to nitrate. Scientists have studied the effect of soil temperature on nitrification rates for decades. Most of this work has been done on soil brought indoors and kept at different temperatures, then adding ammonium and measuring the amount of nitrate that forms over time.
Estimating how quickly ammonium is converted to nitrate in the soil turns out to be complicated, but like most biological processes, it takes temperatures close to freezing to stop the nitrification process completely. Adding the ammonium as anhydrous ammonia also increases the pH and kills a large number of bacteria, effectively delaying the start of nitrification by days or weeks, with longer delays when soil temperatures are lower. Once nitrification begins, soil temperatures in the upper 70s produce maximum rates of nitrification; rates drop to about 50 percent of maximum at 60 degrees, to 10 percent of maximum at 50 degrees, and to about 5 percent of maximum at 40 degrees.
Because nitrification still takes place (slowly) at 50 degrees, why don’t we wait until soil temperature is less than this—say 45 or 40 degrees—before starting to apply ammonia? Waiting until 50 degrees to apply ammonia is a compromise that produces some opportunity to apply at all in falls when harvest is delayed, soils are wet, or soils freeze early. It takes advantage of the delay in nitrification caused by ammonia itself, and of the fact that soil temperatures continue, at least on average, to drop over time. On the other hand, soil temperatures typically rise and fall unpredictably during the fall, and a rise in soil temperature both shortens the delay in the start of nitrification and speeds up the breakdown of nitrification inhibitors.
Managing the risk of having substantial nitrification following application means taking into account both current and predicted soil temperatures when deciding when to apply fall nitrogen. Because soil temperatures fluctuate during the day and are different at different depths, we also need to decide when and at what depth to measure soil temperature. How much the soil temperatures fluctuates during the day depends on soil texture and color, soil moisture, air temperature, and the amount of sunshine. We don’t want to use either maximum or minimum, so often take soil temperature at 10 AM and 4 inches deep under bare soil as an estimate of daily average conditions in the ammonia band after application. Drier soils tend to limit nitrification some, and they also warm and cool more quickly as air temperature changes.
As an example of the challenge of waiting until soil temperature are low enough and likely to stay low, soil temperatures (at 10 AM at a depth of 4 inches under bare soil) averaged over sites in central and northern Illinois as reported by the Illinois Climate Network from October 1 through 25, 2020, are shown in Figure 1 below.
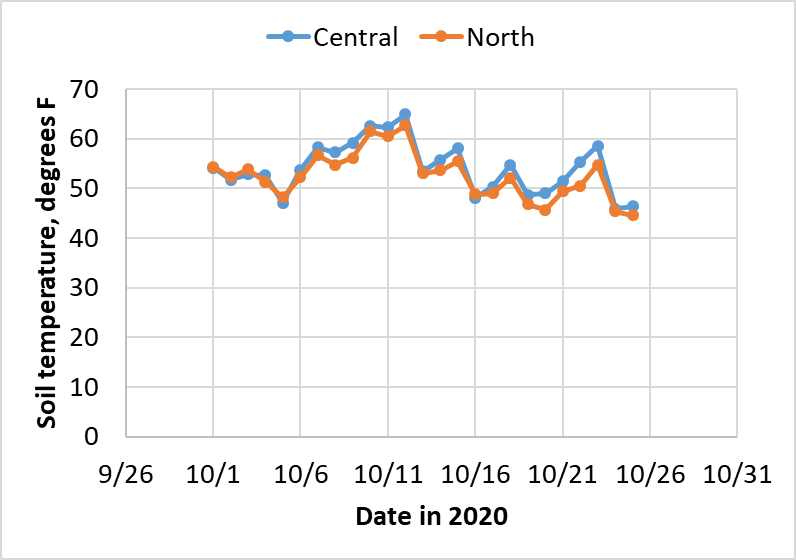
As an exercise, let’s say that we applied 150 lb N as ammonia on the first day that soil temperatures were below 50 degrees (October 5). Let’s further assume that it took 7 days before nitrification kicked in, that the maximum rate of nitrification at high soil temperatures is 10% of the amount applied (15 lb N) per day, and that the nitrification rate rises steadily, from 5% of maximum at 40 degrees to 50% of maximum at 60 degrees. Under these assumptions, 18 lb N per acre in central Illinois and 15 lb N per acre in northern Illinois would have been nitrified by October 25. That might not seem like a lot, but it is in the range of what we measure as tile line nitrate loss following fall (compared to spring) application, and even if temperatures are normal over the next six weeks, more nitrification will take place. With the lag period already over, early application of fall N also means that the nitrification process is primed and ready to go whenever soil temperatures allow.
The good news is that air temperatures have averaged a few degrees below normal in October, and the forecast is that they will remain somewhat below normal this week. Soil temperatures follow air temperatures, of course, and at this point we expect that soil temperatures will drop a little more this week, and that the drop will continue into November. This puts us into the same position we were in late October 2019, when I reported that starting ammonia application at the beginning of the last week of October would not pose an undue risk of nitrification.
We know from records that soil temperatures can rise into the 40s and even into the 50s during the winter, so waiting until soils are cool before applying ammonia doesn’t rule out further nitrification. The fact that periods of heavy rainfall before soils are frozen or after they thaw can result in spikes in surface water nitrates tells us that some nitrification always takes place between fall and spring. Some nitrate that moves from fields is that present in the soil at crop maturity, either from unused fertilizer or N mineralized from soil organic matter after plant uptake stops. But some also comes from fall-applied N; we can minimize this by careful management, and can largely eliminate it by moving application to the spring.
Keep in mind that managing N to minimize loss starts with choosing the best rate. In Illinois that is the MRTN rate, found by using the calculator at http://cnrc.agron.iastate.edu/. It is best to keep some N back to apply in the spring, both to provide flexibility (if prices change) and also to make sure that what is applied in the spring is included in the N rate. If the amount to be applied in the spring is already known—for example, if 45 lb N will go on between herbicide carrier (UAN) and through the planter—then use the expected price for next year’s corn crop and the current price of ammonia in the calculator. At a corn price of $4.00 per bushel and ammonia at $475 per ton ($0.29 per lb N), the MRTN rates are 184 and 191 lb N per acre for corn following soybean in northern and central Illinois, respectively, and 216 and 210 lb N per acre for corn following corn. Besides subtracting the amount to be applied in the spring, subtract also the N applied as MAP or DAP, if those were applied after October 15. If MAP or DAP was applied in September or early October, the N is by now mostly in nitrate form, and the amount subtracted can be decreased by perhaps a third to account for likely loss of some of this.
References:
Nafziger, E. “Is Fall a Good Time to Apply Nitrogen?” Department of Crop Sciences, University of Illinois, September 30, 2020. https://farmdoc.illinois.edu/field-crop-production/is-fall-a-good-time-to-apply-nitrogen.html
Nafziger, E. “Soil temperatures and fall ammonia application.” Department of Crop Sciences, University of Illinois at Urbana-Champaign, October 25, 2019. https://farmdoc.illinois.edu/field-crop-production/uncategorized/soil-temperatures-and-fall-ammonia-application.html





